 Atomic radius patterns are observed throughout the periodic table. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. Test your knowledge across this topic to understand the areas you may need to work. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. A table in which the chemical elements are arranged in order of increasing atomic number. Key Stage 4, Chemistry, Atomic structure and periodic table. The Hampton features a modern contemporary Grey Wood grain finish with heavy-duty quality construction. The Hampton 3-in-1 combination table includes billiards, table tennis, and a Dining table with dual storage bench seating all in one. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). Add the ultimate multi-functional table to your game room that is ideal for the entire family.  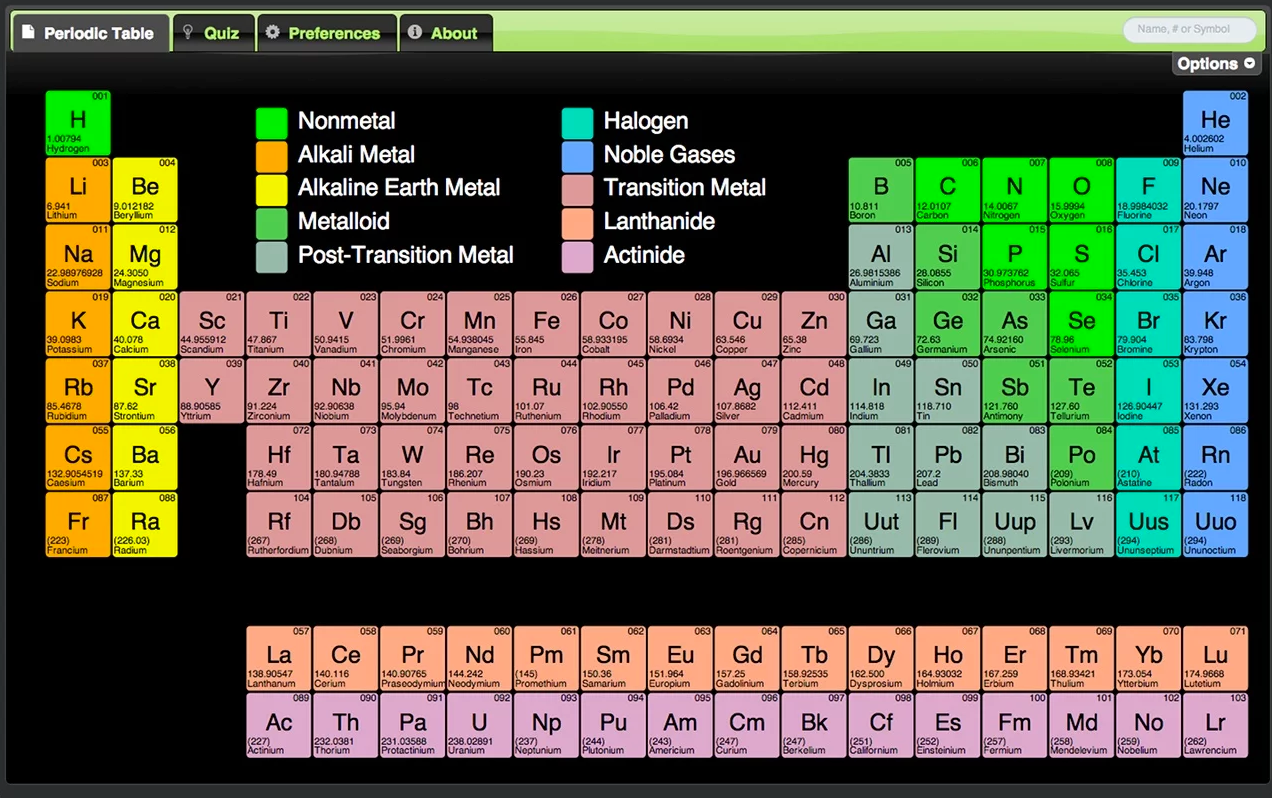
This causes the electron to move closer to the nucleus, thus increasing the electron affinity from left to right across a period. Atomic number of elements steadily increases across a period. The atomic number is how many protons the element’s atom possesses. The elements are ordered by their atomic numbers, which increase as you move across and down the periodic table. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. Read the periodic table from top left to bottom right. Periodic table of the chemical elements The periodic table, also known as the periodic table of the ( chemical) elements, is a tabular display of the chemical elements. Comics include Uncle crooge, Metal Men, Metamorpho, Batman. Moving from left to right across a period, atoms become smaller as the forces of attraction become stronger. Elements in the periodic table are arranged by increasing atomic number (number of protons). Part 1Recognizing the Structure of the Periodic Table. This site contains comic book images linked to the chemical elements via the periodic table. The first 94 elements exist naturally, although some are found only in trace amounts and were synthesized in laboratories before being found in nature.Elements with atomic numbers from 95 to 118 have. All elements from atomic numbers 1 (Hydrogen) to 118 (Oganesson) have been discovered or synthesized. With a larger distance between the negatively-charged electron and the positively-charged nucleus, the force of attraction is relatively weaker. The current Periodic table has 118 elements. This means that an added electron is further away from the atom's nucleus compared with its position in the smaller atom. \( \newcommand\): Periodic Table showing Electron Affinity TrendĮlectron affinity generally decreases down a group of elements because each atom is larger than the atom above it (this is the atomic radius trend, discussed below). 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
